Objective:
The ongoing rFVIIIFc extension study, ASPIRE (clinicaltrials.gov #NCT01454739), evaluates the long-term safety and efficacy of rFVIIIFc in adults, adolescents, and children with severe hemophilia A. Here we report interim outcomes for United States (US) subjects in ASPIRE.
Methods:
Eligible subjects could enroll in ASPIRE upon completing A-LONG or Kids A-LONG. There were 4 treatment groups: individualized prophylaxis (25-65 IU/kg every 3-5 days, or 20-65 IU/kg on D1, 40-65 IU/kg on D4 if twice weekly); weekly prophylaxis (65 IU/kg every 7 days); modified prophylaxis (to further personalize and optimize treatment when needed); or episodic treatment. Subjects could change treatment groups at any time. Subjects <12 yrs participated only in individualized and modified prophylaxis groups. Primary endpoint: development of inhibitors. Secondary outcomes included annualized bleeding rate (ABR) and rFVIIIFc exposure days (EDs).
Summary:
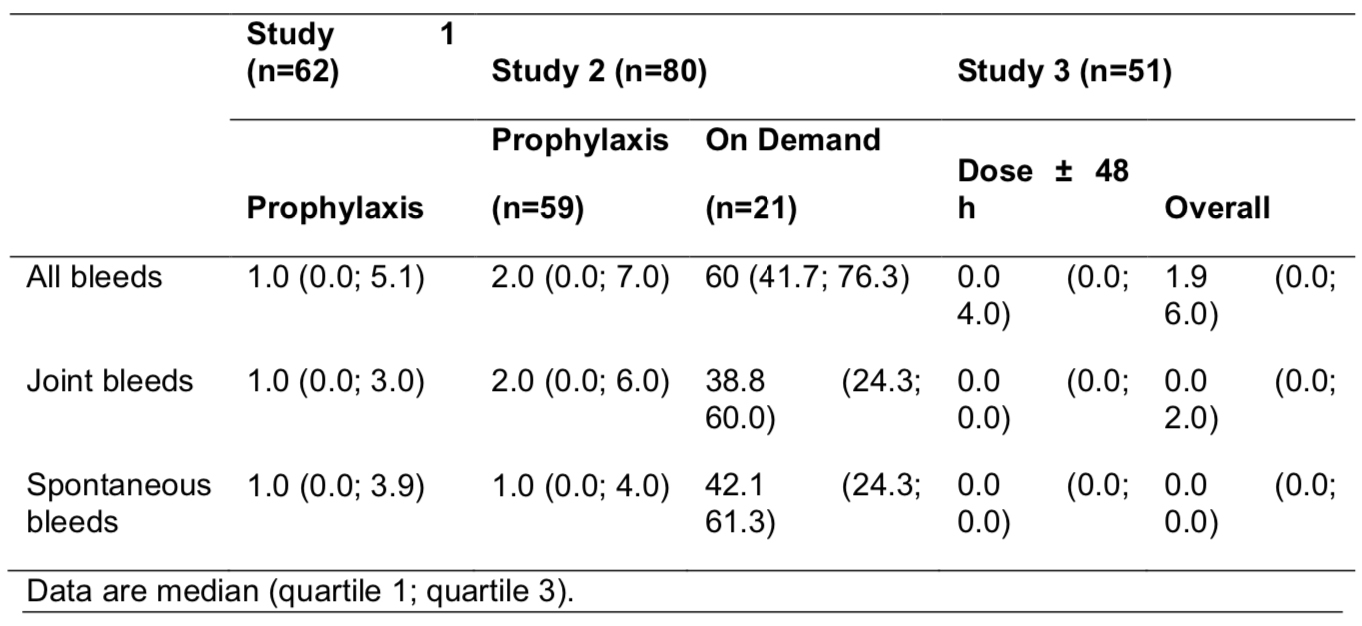
Sixty subjects (49 from A-LONG; 11 from Kids A-LONG) enrolled. As of the interim data cut (6 Jan 2014), the median time on ASPIRE was 80.37 (A-LONG) and 15.94 (Kids A-LONG) wks; 82% (A-LONG) and 27% (Kids A-LONG) of subjects had ≥100 cumulative rFVIIIFc EDs. 7/49 A-LONG subjects changed treatment groups upon enrollment into or during ASPIRE; 2 Kids A-LONG subjects switched from individualized to modified prophylaxis upon enrollment into ASPIRE. Median ABRs were low with rFVIIIFc prophylaxis (Table). Overall, most subjects treated prophylactically during the parent study did not experience changes to their total weekly prophylactic dose or dosing interval during ASPIRE. For subjects who enrolled from A-LONG and Kids A-LONG, 94% and 100% of all bleeding episodes during ASPIRE, respectively, were controlled with 1 infusion. In the overall study population, no inhibitors were observed during ASPIRE; adverse events were typical of the general adult and pediatric hemophilia A populations.
Conclusion:
Interim data from US subjects in ASPIRE are consistent with those of the phase 3 parent studies and the overall ASPIRE interim analysis. Results from ASPIRE confirm the longer-term safety of rFVIIIFc and the maintenance of a low ABR with extended interval prophylactic dosing in individuals with severe hemophilia A.